The Gut Microbiome and Your Health: What the Science Really Says
- Jan 8, 2025
- 10 min read
Updated: Mar 23
By Rita Soares | Functional Medicine Nutritionist | Member of the Portuguese Order of Nutritionists (2604N) & Institute for Functional Medicine
Your gut is home to one of the most complex ecosystems on the planet — and it may be the single most influential factor in your long-term health. From your immune system to your mood, your metabolism to your risk of chronic disease, the trillions of microorganisms living in your intestines are quietly orchestrating far more than digestion.
Yet most people have no idea how directly their daily choices are shaping this ecosystem — for better or worse.
In this article, I'll break down what the gut microbiome actually is, what the current science says about its role in health and disease, and — most importantly — what you can do to support it through food and lifestyle.
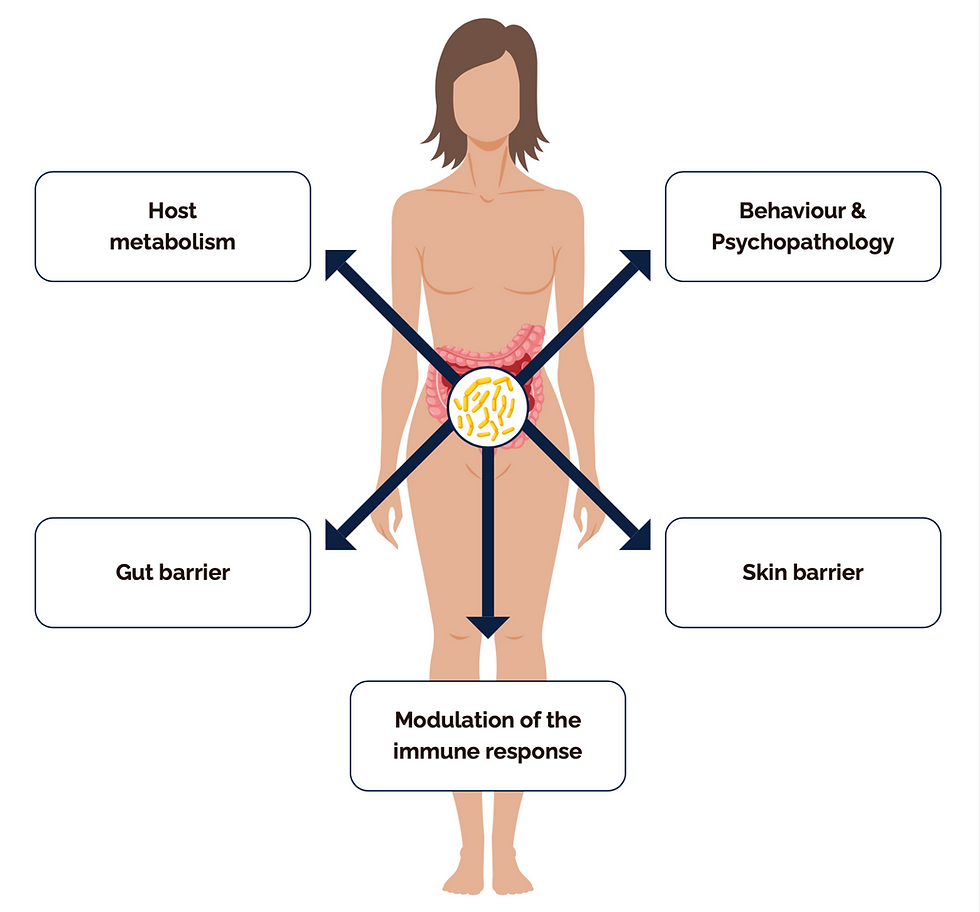
What Is the Gut Microbiome?
The human gastrointestinal tract houses approximately 10 to 100 trillion microorganisms, including bacteria, archaea, fungi, viruses, and protozoa. Collectively, these are known as the gut microbiota. The term microbiome refers to their collective genetic material — a genome estimated to be 150 times larger than the human genome itself (Sender et al., 2016, Cell).
In terms of sheer scale: the gut has a surface area of roughly 200–300 m², and 90% of all gut microorganisms are concentrated in the colon and terminal ileum. Each section of the gastrointestinal tract — stomach, small intestine, large intestine — maintains a distinct microbial community shaped by its unique pH, oxygen levels, and transit time.
The most studied bacterial phyla in the human gut are Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria. A healthy gut is characterised not just by the presence of beneficial species, but by high microbial diversity — the number and variety of species present. Research consistently shows that reduced diversity is one of the strongest markers of gut dysbiosis and systemic disease risk (Lozupone et al., 2012, Nature).
What Does the Gut Microbiome Actually Do?
The functions of the gut microbiome extend well beyond digestion. Here is what the current evidence tells us:
1. Digestion and Short-Chain Fatty Acid Production
Human enzymes cannot break down dietary fibre. Gut bacteria ferment these fibres into short-chain fatty acids (SCFAs)— primarily butyrate, propionate, and acetate. Butyrate is the primary energy source for colonocytes (the cells lining your colon) and plays a critical role in maintaining intestinal barrier integrity and reducing inflammatory signalling (Canani et al., 2011, Journal of Nutritional Biochemistry).
2. Synthesis of Vitamins and Cofactors
Several gut bacteria produce vitamins that humans cannot synthesise in adequate amounts on their own, including vitamin K2, B12, folate, biotin, and riboflavin. Disruption of the microbiome through antibiotics or poor diet can meaningfully reduce these endogenous sources.
3. Regulation of the Immune System
Approximately 70–80% of the immune system is housed in the gut, primarily in gut-associated lymphoid tissue (GALT). Early-life microbial colonisation is critical for training the immune system to distinguish self from non-self, and commensal from pathogenic organisms. Dysbiosis — an imbalance in microbial composition — has been strongly linked to immune dysregulation, autoimmune conditions, and chronic low-grade inflammation (Belkaid & Hand, 2014, Cell).
4. Intestinal Barrier Function
The gut epithelium is a single layer of cells, held together by tight junction proteins. Beneficial bacteria — particularly butyrate-producing species like Faecalibacterium prausnitzii and Akkermansia muciniphila — help maintain tight junction integrity and mucus layer thickness. When these populations decline, intestinal permeability can increase, allowing bacterial endotoxins (lipopolysaccharides) to enter circulation — a phenomenon associated with metabolic disease, neuroinflammation, and chronic fatigue (Leaky gut) (Fasano, 2012, Clinical Reviews in Allergy & Immunology).
5. Metabolic Regulation and Body Weight
The composition of the gut microbiome influences how efficiently energy is extracted from food, bile acid metabolism, and fat storage signalling. Landmark research transplanting microbiota from obese mice into germ-free lean mice induced obesity in the recipients — demonstrating a causal link between microbiome composition and metabolic phenotype (Turnbaugh et al., 2006, Nature). In humans, a higher Firmicutes-to-Bacteroidetes ratio has been associated with increased energy extraction and obesity risk, though this relationship is now understood to be considerably more nuanced.
6. The Gut-Brain Axis
The gut and brain communicate continuously via the vagus nerve, the enteric nervous system, immune signalling, and microbial metabolites. This bidirectional communication system — known as the gut-brain axis — means that the state of your microbiome can directly influence mood, cognition, stress response, and sleep quality.
Gut bacteria produce approximately 90% of the body's serotonin (primarily in enterochromaffin cells, regulated by microbial signals), as well as GABA, dopamine precursors, and neurotrophic factors. Studies in both animal models and humans have linked gut dysbiosis to increased risk of depression, anxiety, and cognitive decline (Cryan et al., 2019, Physiological Reviews).
7. Protection Against Pathogens
A diverse, balanced microbiome provides colonisation resistance — beneficial bacteria occupy available niches, compete for nutrients, and produce antimicrobial peptides (bacteriocins) that inhibit the growth of pathogenic organisms, including Clostridioides difficile and antibiotic-resistant strains.

What Is Gut Dysbiosis and Why Does It Matter?
Dysbiosis refers to a state of microbial imbalance — characterised by reduced diversity, overgrowth of opportunistic or pro-inflammatory species, and depletion of beneficial commensal bacteria.
Dysbiosis has been associated with a broad range of conditions in the research literature:
Gastrointestinal conditions: Irritable Bowel Syndrome (IBS), Small Intestinal Bacterial Overgrowth (SIBO), Inflammatory Bowel Disease (Crohn's disease, ulcerative colitis), and chronic constipation.
Metabolic conditions: Obesity, type 2 diabetes, non-alcoholic fatty liver disease (NAFLD), and insulin resistance.
Immune and inflammatory conditions: Rheumatoid arthritis, Hashimoto's thyroiditis, eczema, psoriasis, and allergies.
Neurological and psychiatric conditions: Depression, anxiety, ADHD, Parkinson's disease, and multiple sclerosis.
Hormonal conditions: Oestrogen dysregulation (via the estrobolome — the subset of gut bacteria that metabolise oestrogens), which has implications for conditions like endometriosis, PCOS, PMS, and perimenopausal symptom severity.
This breadth of association does not mean the microbiome causes all of these conditions — but it does position it as a critical therapeutic target in functional and integrative medicine.
What Disrupts the Gut Microbiome?
Understanding what depletes microbial diversity is as important as knowing how to restore it. The most well-evidenced disruptors include:
Diet: Ultra-processed foods, low fibre intake, excess refined sugar, and high intake of emulsifiers (such as polysorbate 80 and carboxymethylcellulose, found in many packaged foods) have all been shown to reduce microbial diversity and increase intestinal permeability (Chassaing et al., 2015, Nature).
Antibiotics: Broad-spectrum antibiotics can eliminate large segments of commensal bacteria within days. Recovery is possible, but in some individuals — particularly those with pre-existing dysbiosis — the microbiome may not fully return to its original composition without active intervention.
Chronic stress: The hypothalamic-pituitary-adrenal (HPA) axis and sympathetic nervous system directly influence gut motility, secretory IgA production, and microbial composition. Chronic psychological stress has been shown to increase intestinal permeability and shift microbial balance toward pro-inflammatory profiles.
Poor sleep: Sleep fragmentation and circadian disruption alter the composition and metabolic activity of the gut microbiome, creating a bidirectional cycle — dysbiosis also disrupts sleep via serotonin and melatonin signalling.
Sedentary behaviour: Regular physical activity is consistently associated with higher microbial diversity, particularly of butyrate-producing species (Clarke et al., 2014, Gut).
Medications beyond antibiotics: Proton pump inhibitors (PPIs), NSAIDs, oral contraceptives, and metformin have all been shown to alter gut microbial composition.
How to Support Your Gut Microbiome Through Nutrition
This is where clinical practice makes the difference. General advice is rarely enough — the right strategy depends on your symptom profile, diet history, and clinical context. That said, the evidence-based foundations are consistent:
Prioritise Dietary Diversity
Research from the British Gut Project found that eating 30 or more different plant foods per week was the single strongest dietary predictor of microbial diversity — surpassing even probiotic supplementation (McDonald et al., 2018, Cell Host & Microbe). This includes vegetables, fruits, legumes, wholegrains, nuts, seeds, herbs, and spices.
Focus on Prebiotic Fibre
Prebiotics are non-digestible fibres that selectively feed beneficial bacteria. The most studied include:
Inulin and fructooligosaccharides (FOS): Found in garlic, onion, leek, asparagus, chicory root, and Jerusalem artichoke. Feed Bifidobacterium and Lactobacillus species.
Beta-glucans: Found in oats and barley. Support Akkermansia muciniphila and butyrate producers.
Resistant starch: Found in cooled cooked potatoes, green bananas, legumes, and cooled cooked rice. One of the most potent fuels for butyrate-producing bacteria.
Pectin: Found in apples, carrots, and citrus peel.
Include Fermented Foods
Regular consumption of naturally fermented foods has been shown to increase microbiome diversity and reduce markers of immune activation. A 2021 randomised controlled trial from Stanford found that a high-fermented food diet (including yogurt, kefir, fermented vegetables, and kombucha) significantly increased microbial diversity and decreased 19 inflammatory proteins compared to a high-fibre diet alone (Wastyk et al., 2021, Cell).
Practical options: plain live yogurt, kefir (dairy or water), kimchi, sauerkraut, miso, tempeh.
Reduce Ultra-Processed Food Intake
The evidence linking ultra-processed food consumption to gut dysbiosis and increased intestinal permeability is now substantial. Emulsifiers, artificial sweeteners (particularly saccharin and sucralose), and refined carbohydrates all negatively affect microbial composition and the intestinal mucosal layer.
Consider Polyphenol-Rich Foods
Dietary polyphenols are metabolised by gut bacteria into bioactive compounds with anti-inflammatory and antioxidant properties. High-polyphenol foods include extra virgin olive oil, berries, dark chocolate (>75%), green tea, red grapes, and pomegranate. Notably, much of the health benefit attributed to the Mediterranean diet may be mediated through the microbiome.
Hydrate Adequately
The intestinal mucus layer — which protects the gut epithelium and provides habitat for Akkermansia muciniphila — depends on adequate hydration. Aim for 30–35 ml of water per kg of body weight daily.
Probiotics: When Are They Useful?
Probiotic supplements contain live microorganisms intended to confer a health benefit. The evidence is strain-specific and condition-specific — generalised probiotic recommendations are not well-supported by the current literature.
Strains with the strongest clinical evidence for specific conditions include:
Lactobacillus rhamnosus GG: Acute infectious diarrhoea, antibiotic-associated diarrhoea (Szajewska & Mrukowicz, 2001, Journal of Pediatrics)
Saccharomyces boulardii: Prevention of antibiotic-associated diarrhoea and C. difficile recurrence
Bifidobacterium infantis 35624: IBS symptom reduction (Whorwell et al., 2006, American Journal of Gastroenterology)
Lactobacillus acidophilus NCFM: Abdominal discomfort and bloating in IBS
In my clinical practice, I recommend targeted, strain-specific probiotics within the context of a full dietary and lifestyle protocol — not as a standalone intervention.
How I Work with Gut Health in Clinical Practice
As a functional medicine nutritionist with over 11 years of clinical experience and more than 2,000 patients treated, my approach to gut health goes beyond symptom management. In my Gut Health Reset Programme, we:
Identify root causes through detailed clinical assessment and, when appropriate, functional testing (stool microbiome analysis, SIBO breath testing, food sensitivity panels)
Remove dietary and lifestyle triggers actively disrupting the microbiome
Rebuild microbial diversity through a personalised food-first protocol
Reinoculate with targeted, evidence-based probiotic and prebiotic strategies
Repair intestinal barrier function where indicated
Re-balance the nervous system and address stress physiology, which is inseparable from gut health
Most patients begin to notice meaningful improvement within 4–8 weeks. Deeper structural changes in the microbiome — particularly increases in diversity — become apparent over 3–4 months of consistent intervention.
Frequently Asked Questions
What is the difference between microbiota and microbiome? The microbiota refers to the collection of living microorganisms in the gut. The microbiome refers to their collective genetic material and functional capacity. In everyday language, the terms are often used interchangeably.
Can I test my gut microbiome? Yes. Stool microbiome sequencing (16S rRNA or shotgun metagenomics) is available through several commercial laboratories and can provide a detailed overview of your microbial species, diversity indices, and functional capacity. Interpretation should always be done in clinical context — a single snapshot is informative but not definitive.
How long does it take to improve gut microbiome health? Microbial composition can begin to shift within days of dietary change. However, meaningful increases in diversity and stabilisation of a healthier microbial community typically require 8–12 weeks of consistent intervention. Full restoration in cases of significant dysbiosis may take 3–6 months.
Do I need to take probiotics to have a healthy microbiome? Not necessarily. For most people with mild to moderate dysbiosis, a dietary approach focused on diversity, prebiotic fibre, and fermented foods is the primary intervention. Probiotic supplements are most useful in specific clinical contexts — after antibiotic treatment, in IBS, or in conditions with documented depletion of specific species.
Does stress really affect the gut microbiome? Yes — and substantially. Chronic psychological stress activates the HPA axis, increases intestinal permeability, reduces secretory IgA (the gut's first-line immune defence), and shifts microbial composition toward more pro-inflammatory profiles. Managing stress physiology is a non-negotiable part of any gut health protocol.
What foods are most damaging to the gut microbiome? The most consistently harmful are ultra-processed foods (particularly those containing emulsifiers and artificial sweeteners), diets chronically low in fibre, high alcohol consumption, and diets heavily reliant on refined carbohydrates and industrial seed oils.
Is leaky gut a real diagnosis? Increased intestinal permeability — commonly referred to as "leaky gut" — is a real, measurable physiological phenomenon supported by substantial research. It is not yet a standard ICD diagnosis, but it is well-established in the gastroenterology literature and is increasingly recognised as a contributor to systemic inflammation and autoimmune conditions.
Ready to Work on Your Gut Health?
If you're experiencing digestive symptoms, low energy, hormonal imbalances, skin issues, or persistent inflammation — your microbiome may be at the root of it.
In my Gut Health Reset Programme, we go beyond generic advice to identify and address your specific imbalances with a personalised, evidence-based protocol.
Rita Soares is a functional medicine nutritionist based in Lisbon, Portugal, specialising in gut health, longevity, and hormonal balance. Member of the Portuguese Order of Nutritionists (No. 2604N) and the Institute for Functional Medicine (IFM).
Scientific References
Sender R, Fuchs S, Milo R. (2016). Revised estimates for the number of human and bacteria cells in the body. Cell, 164(3), 337–340.
Lozupone CA, et al. (2012). Diversity, stability and resilience of the human gut microbiota. Nature, 489, 220–230.
Canani RB, et al. (2011). Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World Journal of Gastroenterology, 17(12), 1519–1528.
Belkaid Y, Hand TW. (2014). Role of the microbiota in immunity and inflammation. Cell, 157(1), 121–141.
Fasano A. (2012). Leaky gut and autoimmune diseases. Clinical Reviews in Allergy & Immunology, 42(1), 71–78.
Turnbaugh PJ, et al. (2006). An obesity-associated gut microbiome with increased capacity for energy harvest. Nature, 444, 1027–1031.
Cryan JF, et al. (2019). The microbiota-gut-brain axis. Physiological Reviews, 99(4), 1877–2013.
Chassaing B, et al. (2015). Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature, 519, 92–96.
Clarke SF, et al. (2014). Exercise and associated dietary extremes impact on gut microbial diversity. Gut, 63(12), 1913–1920.
McDonald D, et al. (2018). American Gut: an open platform for citizen science microbiome research. Cell Host & Microbe, 23(3), 389–393.
Wastyk HC, et al. (2021). Gut-microbiota-targeted diets modulate human immune status. Cell, 184(16), 4137–4153.
Whorwell PJ, et al. (2006). Efficacy of an encapsulated probiotic Bifidobacterium infantis 35624 in women with irritable bowel syndrome. American Journal of Gastroenterology, 101(7), 1581–1590.




Comments